Peptide regulation of ageing
|
|
||||||||||||||||||||||||||
|
For many years a phenomenon of ageing had been studied within the framework of ethics and social issues. Only in the last century the society realized that a process of ageing should be studied from a different viewpoint, that is, as a special physiologic mechanism bearing a certain evolutionary significance. Ageing is the most complicate issue in medicine and biology. The process of ageing is a gradual involution of tissues and development of organism malfunctioning. Its symptoms appear already at the end of the reproductive period and become more pronounced with ageing. At the end of the XIX century I.I. Metchnikov showed that enhancement of cellular immunity contributed to a lifespan increase. He developed a phagocytic theory of immunity and considered human organism capable of combating pathologic ageing [1]. In 1908 he was awarded the Nobel Prize in Physiology or Medicine together with Paul Ehrlich. And only one century later, Peter Doherty and Rolf Zinkernagel conducted detailed studies in specificity of cellular immunity in case of viral infection (the Nobel Prize in Physiology or Medicine 1996). The development of genetics and molecular biology required profound biochemical studies. Many years of scientific work by Marshall Nirenberg and Gobind Khorana resulted in deciphering genetic code and defining codons (triplets of nucleotides) for each of twenty amino acids (the Nobel Prize in Physiology or Medicine, together with Robert Holley, 1968). Fundamental investigation in nucleic acids biochemistry and identification of RNA and DNA bases sequence were conducted in the 60-70ies of the XX century by Paul Berg, Walter Gilbert and Frederick Sanger (the Nobel Prize in Chemistry, 1980). Experimental and clinical studies in gerontology evidenced that immunity of the organism is among the first systemic functions to reveal disorders with ageing [2, 3]. Thymus peptide extracts and peptides, isolated from these extracts, were the first preparations suggested for immune deficit correction [4, 5, 6]. The origin of small regulatory peptides pool in a young organism became evident after the discovery of ubiquitin-mediated protein degradation in proteasomes made by Aaron Ciechanover, Avram Hershko, Irwin Rose (The Nobel Prize in Chemistry, 2004). Small peptides were shown to play an important role for transmission of biologic information, as for example autocrine hormones and neuropeptides do. A high molecular protein can be hydrolyzed in different ways, degradating into several small peptides. Due to this mechanism there can be produced peptides with completely different biologic functions as compared to the maternal macromolecule [7]. American mathematician Samuel Karlin proved in his works that there are several types of recurring blocks of amino acid residues with charged side chains in protein macromolecules. Structures of transcription factors reliably contain the greatest number of such blocks [8]. In 1961 Francois Jacob and Jacques Monod suggested a model of genetic regulation of protein synthesis with the participation of low-molecular ligand, which ousts repressor and triggers allosteric conformational transition in the DNA structure of bacterial cell [9]. In 1965 they were awarded the Nobel Prize in Physiology or Medicine along with Andry Lwoff. However, the regulating role of small peptides had never been discussed in the concepts of gene control of protein synthesis in higher organisms before we started the research. Alongside with immunity decrease associated with age there occur other alterations at cellular level. In particular there take place changes in the inner structure of cellular nucleus. DNA-protein complex of cellular nucleus (chromatin) organizes into chromosomes only in case of cell division. In stationary state chromatin is found in two forms: euchromatin and heterochromatin [10]. Heterochromatin is usually localized in the nucleus periphery and contains generally inactive part of genome: genes blocked by repressors. The ratio euchromatin/heterochromatin changes with ageing due to reduction of active euchromatin. This leads to the decrease of protein synthesis in a cell [11]. Thus ageing phase of the organism reveals several levels of disfunction and may be classified as a systemic syndrome. Promising results of immunodeficiency correction by endogenous regulatory peptides testified the necessity of further enhanced studies [4,5]. |
||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||
|
It is known that specific limit of animal and human lifespan is approximately 30-40% higher than their mean lifespan. It could be referred to the impact of adverse factors causing changes in the gene structure and expression accompanied by disorders in the protein synthesis and organism functioning (Fig. 1). |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 1. Potential increase in the average human lifespan up to the specific limit (biological reserve). | ||||||||||||||||||||||||||
|
At the beginning of the 1970ies we studied the mechanism of immunodepression both experimentally and clinically. We found out that with ageing there takes place an involution of the central organ of immune system – thymus (Fig. 2, 3) and that of the neuroendocrine system - pineal gland. We also registered definite decrease of protein synthesis in cells of different organism tissues (Fig. 4). |
||||||||||||||||||||||||||
 |
Subcapsular cortex area (open biopsy, a 2-year old child). A - hematoxylin and eosin staining; B - thymic polypeptides fluorescence in the bodies and processes of epithelial cells forming Klark's alveoli, as well as granules on the thymocyte membranes inside alveoli. |
|||||||||||||||||||||||||
 |
Subcapsular cortex area (section biopsy, a 46-year old man). A - hematoxylin and eosin staining; B - thymic polypeptides fluorescence in the bodies and processes of epithelial cells forming groups of 2-5 cells. |
|||||||||||||||||||||||||
| Fig. 2. Age-related involution of thymus (indirect immunofluorescent method with antibodies to thymic polypeptides, x600). | ||||||||||||||||||||||||||
 
|
Immuno fluorescence laser confocal microscopy, х400 (red fluorescence - Rodamin G, green fluorescence - FITC). |
|||||||||||||||||||||||||
| Fig. 3. Expression of transcription proteins (PAX 1) in human thymus epithelial cells (the study was conducted in cooperation with Prince Philippe Biomedical Research Center, Valencia, Spain). | ||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 4. Protein synthesis in hepatocytes of rats of different age. | ||||||||||||||||||||||||||
|
To restore functions of thymus, pineal gland and other organs we developed a special method for isolation, refinement and fractionating of low-molecular peptides from extracts of these organs [12, 13]. Low-molecular peptides isolated from thymus (preparation Thymalin) and pineal gland (preparation Epithalamin) of animals were studied in different biologic models. These peptide preparations contributed to a reliable increase in animals mean lifespan in numerous experiments (Fig. 5) [12, 13, 14, 15, 16, 17]. Of particular importance is a correlation between mean lifespan and the main index of cellular immunity (reaction of blast-transformation of lymphocytes with phytohaemagglutinin) determining T lymphocytes function (Fig. 6) [14]. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 5. The increase in the average lifespan up to the specific limit after peptide preparations application (mean results after 15 experiments). | ||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 6. Peptide preparations effect on mean lifespan and reaction of blast-transformation of lymphocytes with phytohaemagglutinin in mice. | ||||||||||||||||||||||||||
|
A significant increase in the mean lifespan of animals was evidently caused by a reliable antitumoral activity (Fig. 7) of low-molecular peptides isolated from thymus and pineal gland [14, 15, 17, 18, 19]. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 7. Effect of pineal gland preparation on the frequency of tumor incidence in animals. | ||||||||||||||||||||||||||
|
Small peptides isolated from different organs and tissues as well as their synthesized analogues (di-, tri-, tetrapeptides) revealed a realiable tissue specific (gene specific) effects both in cellular cultures and in experimental in young and old animals (Fig. 8). |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 8. Peptide tissue-specific (gene-specific) regulation. | ||||||||||||||||||||||||||
|
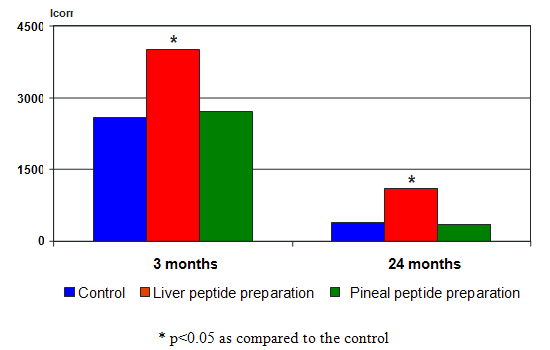
Peptide tissue specific activity manifested in stimulation of protein synthesis in cells of those organs they had been isolated from. The enhancement of protein synthesis under the effect of peptide has been registered in young and old animals (Fig. 9). |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 9. Effect of the liver and pineal peptide preparations on protein synthesis intensity in hepatocyte monolayer culture in rats of different age. | ||||||||||||||||||||||||||
|
Especially significant appeared restoration of reproductive function system in old female rats subjected to the pineal peptide treatment (Fig. 10) [13]. Estrus phase in animals, analogous to menopause in women, lowered from the initial 95% down to 52% after the preparation administration, while other phases of the cycle, typical of the normal estrus, increased from the initial 5% up to 48%. It should be emphasized that in a special experiment initially none of the rats got pregnant after mating. Repeated mating after the administration of the pineal gland peptide entailed pregnancy in 4 out of 16 animals which gave birth to 5-9 healthy off-springs each. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 10. Effect of peptide preparation of the pineal gland on constant estrus in old female rats. | ||||||||||||||||||||||||||
|
Thus there were ascertained main advantages of low-molecular peptides: they possessed high biologic activity, revealed tissue specificity and were neither species specific nor immunogenic. These features make regulatory peptides similar to peptide hormones [16, 20, 21]. A detailed study of molecular weight, chemical properties, amino acid composition and sequence of low-molecular peptides isolated from thymus, pineal gland and other organs had been carried out for many years [22, 23, 24, 25, 26]. The obtained data were used for chemical synthesis of several small peptides. A comparative analysis showed that biological activity of natural and synthetic preparations was largely identical. Thus, for example, thymus dipeptide stimulated immunity [24, 25]. Biological activity of natural and synthetic peptides appeared to be similar in standard testing in tissue cultures and in animals [27, 28, 29, 30]. These results demonstrate prospects for application of these peptides as geroprotectors [31, 32, 33]. The necessity of searching for new drugs – geroprotectors dictated the onset of preclinical studies of these preparations on different structural levels. On the level of the organism in different animals we have registered a significant variety of biologic effects exerted by small peptides especially by peptides of thymus and pineal gland, including proliferative activity and apoptosis [34, 35, 36, 37, 38, 39, 40, 41, 42]. On the level of cellular structures, small peptides activate heterochromatine in the cell nuclei in senile patients (Table 1) [11, 43]. |
||||||||||||||||||||||||||
|
Table 1
Effect of peptides on chromatin in lymphocytes of old humans
** p<0.001 compared to the control (75-88 years old) |
||||||||||||||||||||||||||
|
As it was said above there are two forms of chromatin in the cellular nucleus: light euchromatin and dense heterochromatin located near nuclear membrane. Gene transcription takes place in the light phase, that's in euchromatin. With ageing the amount of heterochromatin in the nucleus increases on average from 63% to 80%. Regulatory peptides entail the increase in the amount of euchromatin in the nucleus. This means that more genes become available for transcription factors, and transcription of gene information goes on more intensively as well as protein synthesis. In other words the more euchromatin there is in the nucleus the more intensive the protein synthesis in the cell is [10, 11, 43]. The capability of peptides to induce polypotential cells differentiation is of special significance (Fig. 11) [42]. Thus addition of retinal peptides to polypotential cells of Xenopus laevis early gastrula ectoderm led to the emergence of retinal and pigment epithelium cells. This outstanding result explains a pronounced clinical effect of the preparation of the retina in patients with retinal degenerations [42] and in animals with genetically determined retinitis pigmentosa. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 11. Induction effect of retinal peptides on the polypotent cells of Xenopus laevis early gastrula ectoderm. | ||||||||||||||||||||||||||
|
On a chromosome level the number of chromosomes aberrations was used a marker of DNA damages in an ageing organism. Somatic mutations can occur due to accumulation of stable aberrations and underlie age-related pathology, including malignant neoplasia. Reliable antimutagenic and reparative activity of thymus and pineal gland peptides have been confirmed by a reduction in the number of chromosome aberrations in the bone marrow cells and cornea epithelium cells in animals revealing accelerated ageing [45]. On the level of gene activity regulation it was established that administration of peptides Lys-Glu and Ala-Glu-Asp-Gly to transgenic mice caused a 2–3.6-fold suppression of HER-2/neu gene expression (human breast cancer) as compared to the control group. This suppression is accompanied by a reliable reduction of the tumor diameter (Fig. 12) [41]. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 12. Peptides effect on the development of mammary adenocarcinoma and HER-2/neu oncogene expression in transgenic mice (the study was conducted in cooperation with the National Research Centre on Ageing, Ancona, Italy). | ||||||||||||||||||||||||||
|
It was revealed that addition of tetrapeptide Ala-Glu-Asp-Gly to the cultural medium of human lung fibroblasts induces telomerase gene expression and contributes to a 2.4-fold lengthening of telomeres. Activation of gene expression is accompanied by a growing number of cellular divisions (by 42.5%), which is the evidence of Hayflick's limit overcoming (Fig. 13) [46, 47]. The effect of di- and tetrapeptides Lys-Glu, Glu-Trp, Ala-Glu-Asp-Gly, Ala-Glu-Asp-Pro on the expression of 15 247 murine heart and brain genes before and after peptides administration was studied with the employment of DNA-microarray technology [48]. In this experiment, there were used clones from the library of the National Institute on Ageing, USA. This experiment provided unique data on alteration in the expression of different genes under the effect of peptide preparations (Fig. 11). An important conclusion driven from the experiment was that every peptide specifically regulates particular genes. Results of this experiment testify to the existing mechanism of peptide regulation of gene activity. It was also registered that dipeptide Lys-Glu, showing immunomodulating activity, regulates gene interleukin-2 expression in blood lymphocytes [49]. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 13. Overcoming human somatic cell division limit due to introduction of the peptide into the culture of the pulmonic fibroblasts. | ||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 14. Peptide effect on gene expression in the heart of mice (the study was conducted in cooperation with the National Institute on Aging, Baltimore, USA). | ||||||||||||||||||||||||||
|
On the molecular level, there was an obvious gap between multiple evidence of specific effects, caused by regulatory peptides in activation of gene transcription [50, 51, 52, 53, 54, 55, 56, 57, 58], and limited schemes of the process underlying the selective binding of the transcription factor with specific DNA sites. Meanwhile non-specific binding of proteins with the DNA double helix was proved using physicochemical methods [59]. Activation of gene transcription in cells of higher organisms as a rule needs dozens of macromolecular activators and transcription factors. We proposed a molecular model of interaction between regulatory peptides and DNA double helix in gene promoter region of (Fig. 15, 16, 17, 18) [60, 61]. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 15. Unfolded peptide Ala-Glu-Asp-Gly conformation (plan projection). There are shown end and side functional groups, capable of complementary interaction with DNA. —NH3 - proton donors groups; =O – proton acceptors groups; —CH3 – hydrophobic (methyl) group. Bold line - main peptide chain. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 16. Metric location of functional groups exposed onto the surface of the DNA major groove in case of embedding of each nucleotide pair into DNA double helix. Dash line - perpendicular plane, where aromatic structures of nucleic bases are located. —NH2 - proton donors groups; =7N – proton acceptors groups; —CH3 – hydrophobic (methyl) group. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 17. Sequence of nucleotide pairs in the DNA double helix, functional groups of which are complimentary to functional groups of Ala-Glu-Asp-Gly peptide. This sequence is many times repeated on the promoter segment of telomerase gene. | ||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 18. Scheme of complementary interaction of tetrapeptide Ala-Glu-Asp-Gly with DNA double helix (“DNA-tetrapeptide” complex on the promoter segment of telomerase gene). | ||||||||||||||||||||||||||
|
Geometrical and chemical complementarity of peptide amino acid sequence and DNA nucleotide pairs sequence was assumed as a basis for the molecular model. Regulatory peptide recognizes a specific site in the DNA double helix if its own amino acid sequence is complementary to the DNA nucleotide sequence for a sufficient length. In other words their interaction is specific due to matching sequences. Each sequence of the DNA double helix nucleotide pairs forms a unique pattern of functional groups on the surface of the DNA double helix major groove. A peptide in the unfolded β-conformation can complimentary fit into the in the DNA major groove along the double helix axis. We used data on molecular geometry of the DNA double-helix and peptide β-thread from scientific publications in order to identify nucleotide pairs sequence for specific binding of the DNA and peptide Ala-Glu-Asp-Gly. The screening conducted showed that this tetrapeptide can be located in the DNA major groove with the ATTTG (or ATTTC) nucleotide sequence on the main chain in compliance with the complementarity of disposition of their functional groups [59]. For experimental testing of the molecular model there were used synthetic preparations: DNA [poly(dA-dT):poly(dA-dT)] (double helix) and tetrapeptide Ala-Glu-Asp-Gly. Gel chromatography helped to prove that peptide Ala-Glu-Asp-Gly forms stable intermolecular complex with the DNA double helix (Fig. 19) [61]. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
|
1 - individual Ala-Glu-Asp-Gly peptide
Fig. 19. HPLC of peptide and DNA on sefadex G-25 in physiological solution at room temperature.
2 - free DNA double helix [poly (dA-dT) : poly (dA-dT)] 3 - mixture of peptide and DNA |
||||||||||||||||||||||||||
|
Complementary binding of the peptide with nucleotides sequence on the leading strand TATATA of the double helix can be conducted by six hydrogenous and one hydrophobic bonds between functional groups of the both participants. Under normal physiological conditions DNA exists in the form of a double helix two polymeric chains of which are kept together by hydrogenous bindings between pairs of bases of each chain. Most of the biological processes with DNA participation (transcription, replications) need the double helix to undergo disjunction into separate strands. In particular, it is known, that local separation of double helix strands precedes gene transcription by RNA polymerase. For the transcription onset (synthesis of the matrix RNA) the DNA double helix has to be freed from histones, and in the place where the matrix RNA synthesis starts, the strands of the double helix should be disintegrated (Fig. 20). |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 20. The scheme of local separation of strands [poly (dA-dT) : poly (dA-dT)] as a result of peptide Ala-Glu-Asp-Gly binding in DNA double helix major groove. | ||||||||||||||||||||||||||
|
Concentration dependent hyperchromic effect (increased optic density 260 nm) was found by spectrophotometery of solutions containing synthetic DNA double helix and tetrapeptide Ala-Glu-Asp-Gly. The hyperchromic effect points out a partial destruction of hydrogen bonds between nucleotide pairs of the double helix and local separation of its strands (allosteric conformational changes) [61]. It was experimentally revealed that detachment (melting) of free DNA strands occurs at the temperature +69.50С. In the “DNA-tetrapeptide” system melting of the double helix occurred at the temperature +28°С and was characterized by approximately 2-fold decrease in the values of entropy and enthalpy. This fact points out a thermodynamically simplified way of the DNA strands separation in temperature settings typical of biochemical processes of the majority of living organisms. In vitro experiments show that a small peptide of the definite structure and amino acid sequence can participate in activation of genes transcription on the stage of strands disjunction in the DNA double helix. Biochemical aspect of this phenomenon consists in similarity of structure and amino acid sequence of a regulatory peptide and a specific segment of the peptide chain of the macromolecular transcription factor. Thus, the studies of peptides biological activity on different structural levels and of physicochemical processes of their interaction proved an indubitably high physiologic activity of peptide regulators. Major conclusion reads that peptides are capable of regulating gene expression. Pre-clinical trials demonstrated high biological activity and safety of synthesized peptides [61, 63, 64, 65]. Thus, the administration of peptides Lys-Glu, Ala-Glu-Asp-Gly to animals contributes to a reduced incidence of tumors and an increase of mean lifespan [66, 67, 68]. Peptide Ala-Glu-Asp-Pro stimulates nerve regeneration [69], peptide Lys-Glu-Asp-Trp decreases blood glucose level in animals with experimental diabetes mellitus [70]. Taking into consideration a reliable biological activity of peptides we found it reasonable to study the effect of regulatory peptides in monkeys. Restoration of the melatonin level up to normal following the administration of the peptide preparation to old monkeys was among our significant achievements. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 21. The peptide effect on melatonin production in monkeys of different age. | ||||||||||||||||||||||||||
|
The same old monkeys revealed a restoration to normal indices of a daily rhythm of secretion of the main hormone of adrenal gland – cortisol (Fig. 22). |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 22. The peptide effect on cortisol production in monkeys of different age (in the morning and in the evening). | ||||||||||||||||||||||||||
|
Taking into consideration the encouraging data testifying to high geroprotective activity of both natural tissue specific and synthetic peptide preparations we have been concentrating our attention on studies of geroprotective activity of peptides in old and senile people in recent years [42, 57, 71, 72, 73, 74, 75, 76]. Thus, annual treatment course with thymus and pineal preparations led to a reliable decrease in mortality (Table 2), due to improvement of brain function and that of immune, endocrine, cardio-vascular systems, increased density of osseous tissue (Fig. 23, 24) [42, 57, 71, 72]. It is noteworthy that application of preparation of the thymus led to a 2-fold decrease in frequency of acute respiratory disease (Fig. 25) [57]. The restoration of melatonin secretion level in patients subjected to administration of preparation of the pineal gland is of special significance (Fig. 26) [71, 71]. These results suggest good prospects for tackling demographic issues [77]. |
||||||||||||||||||||||||||
|
Table 2
Peptide preparation effect on mortality rate in elderly and old patients
|
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 23. Effect of thymus preparation on metabolism in elderly patients (60-74 y.o.). | ||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 24. Dynamics of reaction of blast-transformation of lymphocytes with phytohaemagglutinin index in elderly patients in 3 years after 6 courses of peptide preparations. | ||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 25. ARD incidence in elderly and old people treated with thymus preparation. | ||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 26. Effect of the pineal gland preparation on melatonin level in elderly people. | ||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||
|
The mechanisms of ageing studies showed that an involution of the main organs and tissues of the organism accompanied by a decrease of protein synthesis in cells underlies the process of ageing. Peptide preparations isolated from organs of young animals when introduced into an old organism are capable of inducing protein synthesis, followed by restoration of the main functions. It was registered that long-term application of peptides, both isolated from the organs and synthesized from the amino acid, in animals (as a rule starting from the second half of their life) leads to a reliable increase in their mean lifespan by 20-40% and reaching a specific limit. It was revealed that small peptides (di-, tri- and tetrapeptides) are capable of complementary interaction with the DNA specific binding site on the promoter segment of genes, inducing disjoining of double helix strands and RNA polymerase activation. Discovery of the phenomenon of peptide activation of gene transcription points out the natural mechanism of organism to maintain physiologic functions, which is based on the complementary interaction of the DNA and regulatory peptides. This process is fundamental for the development and functioning of the living substance (Fig. 27, 28), while ageing is an evolutionary determined biologic process of age-related changes in gene structure and expression. Application of peptide bioregulators in humans for preventive purposes led to a significant rehabilitation of the main physiological functions and a reliable mortality decrease in different age groups during the period of 6-12 years. |
||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 27. The role of peptides in the cycle of DNA, RNA and protein biosynthesis. | ||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
| Fig. 28. Mechanism of peptide regulation of the living matter development. | ||||||||||||||||||||||||||
|
It should be emphasized that this approach to the prevention of ageing is based not only on experimental and clinical data, but also on technological developments having world novelty [78, 79, 80, 81, 82]. The author and his group believe that the whole complex of 35-year experimental and clinical studies may serve a vital contribution to the advancement of a scientific heritage of the prominent Russian scientist I.I. Mechnikov in the field of gerontology and will be to the benefit of people, especially for those of old and old old age. |
||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||
|
The author expresses his sincere gratitude to: Academicians of the Russian Academy of Sciences and of the Russian Academy of Medical Sciences A.I. Grigoriev, M.A. Paltsev, R.V. Petrov; Academicians of the Russian Academy of Sciences V.T. Ivanov, S.G. Inge-Vechtomov, A.D. Nozdrachev; Academicians of the Russian Academy of Medical Sciences V.G. Artamonova, I.P. Ashmarin, N.P. Bochkov, F.I. Komarov, E.A. Korneva, B.A. Lapin, G.A. Sofronov, K.V. Sudakov, B.I. Tkachenko, V.A. Tutelyan, Academicians of the Academy of Medical Sciences of Ukraine O.V. Korkushko and G.M. Butenko, Associate Members of the Russian Academy of Sciences D.P. Dvoretskiy, Associate Members of the Russian Academy of Medical Sciences G.M. Yakovlev, Professors V.N. Anisimov, A.V. Arutjunian, B.I. Kuznik, L.K. Shataeva, researchers of the Saint Petersburg Institute of Bioregulation and Gerontology of the North-Western Branch of the Russian Academy of Medical Sciences Professors I.M. Kvetnoy, V.V. Malinin, V.G. Morozov, G.A. Ryzhak, distinguished doctor of the Russian Federation L.V. Kozlov, doctors E.I. Grigoriev Ph.D., S.V. Anisimov Ph.D., I.E. Bondarev Ph.D., O.N. Mikhailova Ph.D., A.A. Chernova, and foreign colleagues Professors T.A. Lezhava (Georgia), A.I. Yashin (USA), J. Atzpodien (Germany), K.R. Boheler (USA), C. Franceschi (Italy), E. Lakatta (USA), J. Martinez (France), M. Passeri (Italy) for their significant contribution into our research work. |
||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
